Cell line - HEK293T
HEK293T is a human embryonic kidney cell line. Images have been generated with BioTek Cytation5 under environmental control (37 ℃ and 5% CO2) in one field of view. The cells were maintained in a BioTek Biospa8 automated incubator (37 ℃ and 5% CO2) during the interval period of imaging. Cells had the nuclear reporter H2b-mRuby.
Cell lines were seeded onto 12 well plates (Corning Falcon #353043). Collagen type I (Corning #354236) coated (5µg/cm2) plates were used to facilitate better adhesion of the cells to the bottom of the wells. The seeding density were 3-4×104 cells/well. Cells were incubated for 16 hours at 37 ℃ and 5% CO2, then the medium was changed to remove the dead cells and further incubated for 2 hours. Images were acquired at 4X, 10X and 20X magnifications with RFP channel (531/593) to visualize red fluorescent protein, mRuby as well as Phase contrast channel with 10-15 minutes interval for three days.
HEK293T cells expressing H2b-mRuby were cultured in DMEM (Gibco #10313021) supplemented with 10% FBS (V/V) (Gibco #10082139), 2mM L-glutamine (Corning #25-005-CI) containing 0.5 µg/mL puromycin (STEMCELL #73342). For trypsinization 0.25% Trypsin-EDTA (Gibco, #25200-072) was used for HEK293T cells.
Generation of HEK293T Cell Line Expressing H2b-mRuby
Electroporation was used to deliver the pLenti-Puro-H2b-mRuby plasmid to the HEK 293T cells (ATCC, CRL-3216). HEK 293T cells (passage 4) were trypsinized (0.25% Trypsin-EDTA, Gibco #25200-072), centrifuged (100g, 5mins, RT), and resuspended in full growth medium. 1×104 cells were transferred to a pre-chilled 1.5 mL tube, and centrifuged again (300g, 5mins, 4℃). The cell pellet was resuspended in 400 µL Opti-MEMTM (Gibco #51985091) and aliquoted into two pre-chilled 0.2 cm electroporation cuvettes (Bio-Rad #165-2086) for the autoclave Milli-Q water (negative control) and 10 µg of pLenti-Puro-H2b-mRuby plasmid (sample).
Electroporation was performed using Gene Pulser XcellTM (Bio-Rad) with the preset HEK 293T protocol: Square Wave, 110V, 25.0 msec pulse, 0.2 cm gap cuvette, 200 µL (5×105 cells) per cuvette. Cells were immediately transferred to a 6-well plate (Corning, Falcon #353046) with 1.8 mL full growth medium, with media changed every 24 hours to remove dead cells.
At 96 hours post-electroporation, when confluency reached to 90%, cells were selected with puromycin (1 µg/mL, STEMCELL #73342) for three days until all negative control cells were dead, and those sample cells reached 90% confluency. Stable H2B-mRuby expressing cells were maintained in full growth medium with 0.5 µg/mL puromycin.
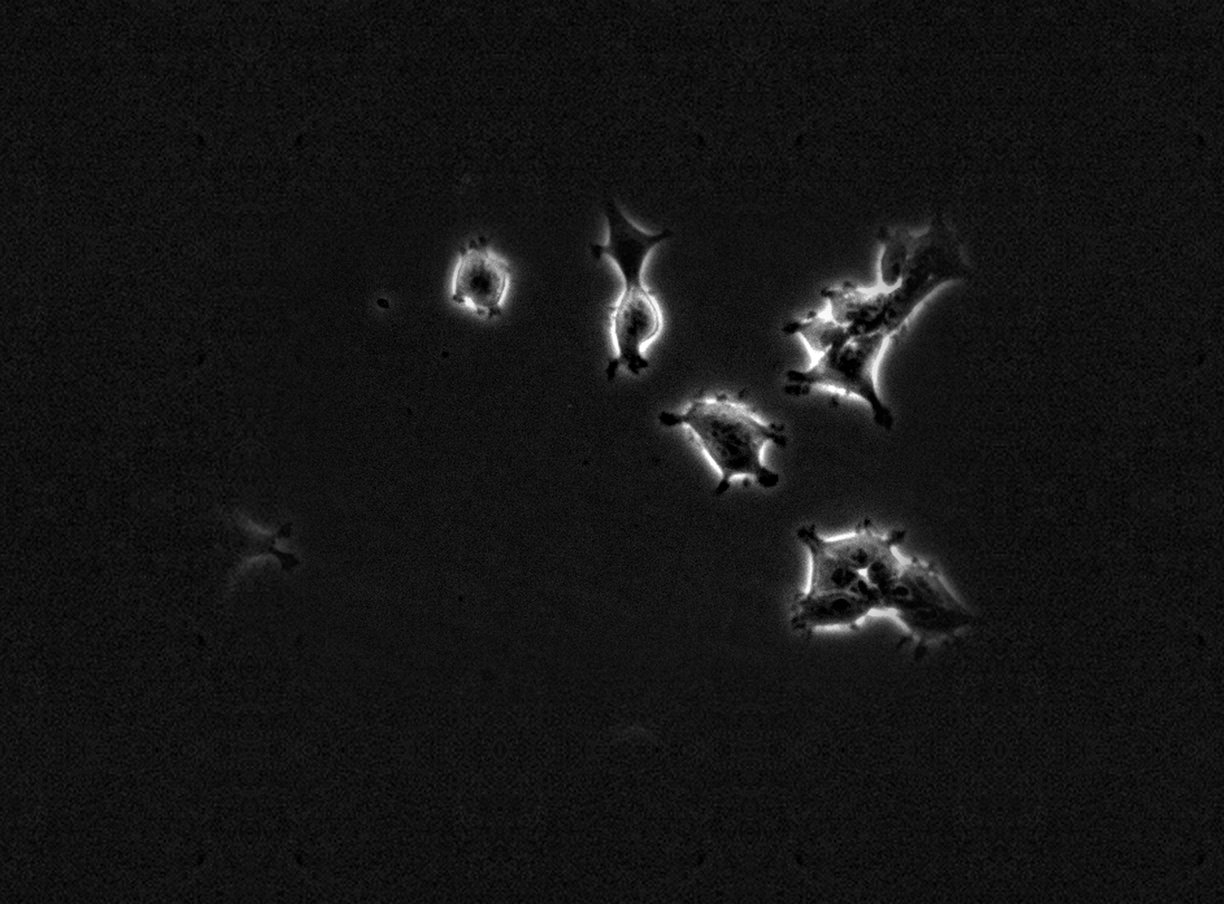

FLD_1
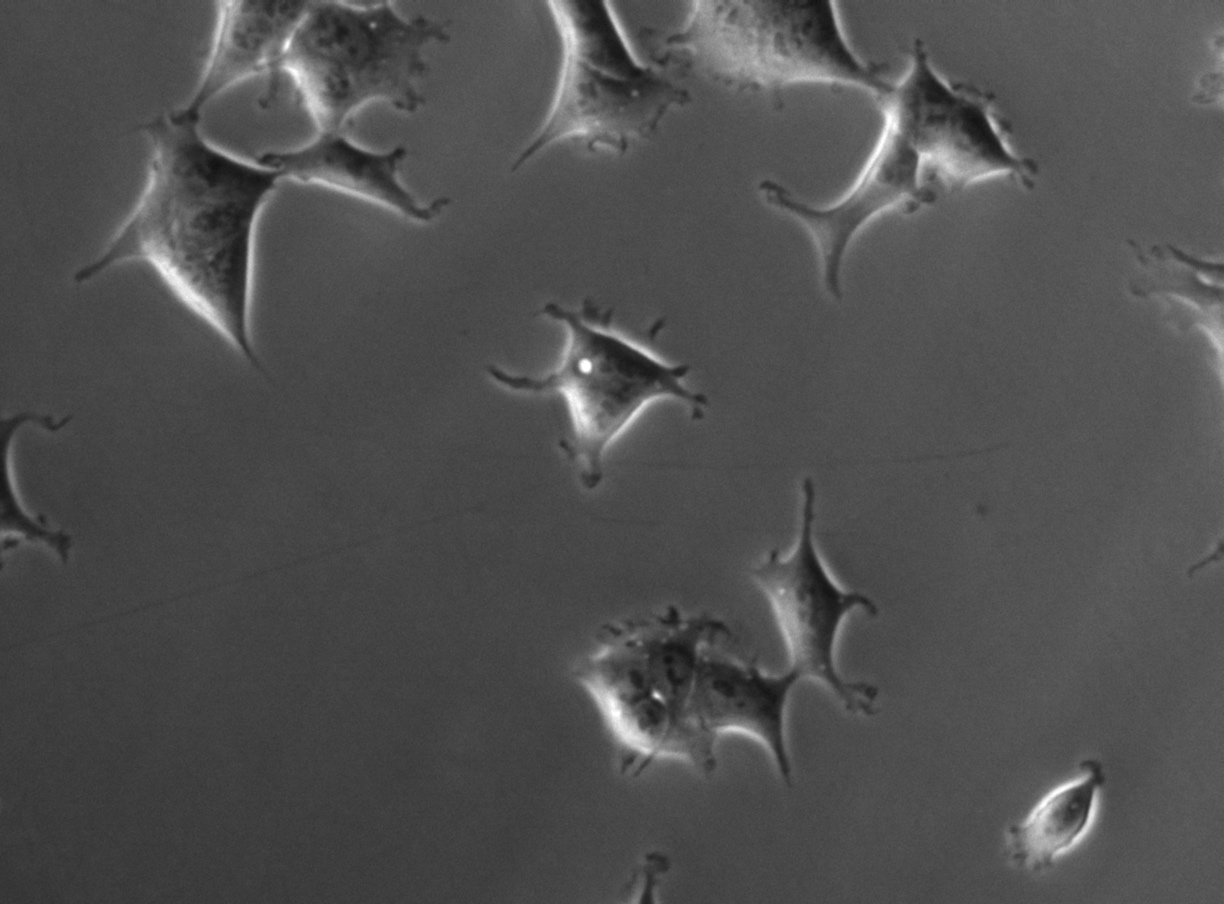

FLD_14
Example sequences from the HEK293T dataset. Left: phase contrast. Right: nuclear fluorescence (RFP).
Images
Annotation
Videos
10× magnification
FLD_1
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_2
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_3
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_4
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_5
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_6
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_7
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_8
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
20× magnification
FLD_2
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_5
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_8
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_14
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_23
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation
FLD_29
GAP 1: pc | rfp | annotation
GAP 2: pc | rfp | annotation
GAP 4: pc | rfp | annotation
GAP 8: pc | rfp | annotation